Research Interests
There is compelling evidence that chemistry
is an important contributor the non-sustainable direction in
which the planet is heading. Chemistry MUST be part of the
solution. Our hypothesis is that silicones can be a part of a
sustainable future.
Silicones undergo degradation in the environment through
hydrolysis, oxidation and microbiological processing to give
CO2, water and sand. To improve sustainability, we
need to reduce the energy content of silicones for a given
application, use more natural feedstocks, and better design
for (bio)degradability; this would align silicone polymers
with the 12 rules of Green Chemistry. Our approaches are
summarized below.
Nature provides many different products that
can serve as starting materials for silicones: lipids (255,
302),
proteins (145),antioxidants,
including vitamins, and saccharides. Some of these can simply
act as diluents that should improve degradation in the
environment. More interesting is the ability to deliver new
function to silicones, including antioxidant properties (300),
artificial enzymes (294) and
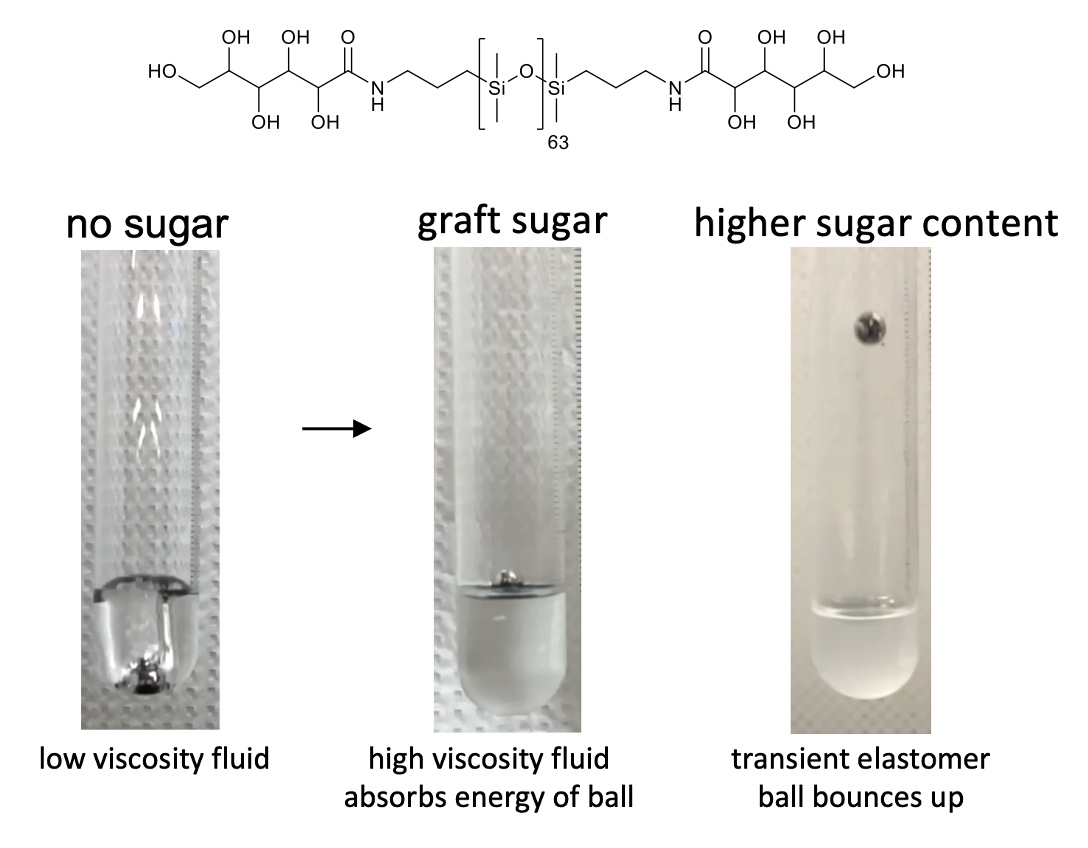
more force responsive, thermoplastic elastomers based on sugar
(268).
B. Catalysts are Great (Green Chemistry
Rule 9); Would Avoidance Be Better?
Catalysts can dramatically reduce both inputs and waste
produced. However, they can also be problematic do to cost or
toxicity; silicone elastomers are frequently formed using tin
and platinum catalysts. Less problematic catalysts include
bleach (282),
peroxide, or photogenerated radicals to generate (in some
cases, recyclable thermosets (252,
271).
Alternatively, simple organic chemistry leads to thermosets
from click reactions based on the Huisgen cyclization (244),
dynamic, thermoplastic elastomers using imine
bonds (267, 289),
or even simple ionic bonds (275).
We continue to explore simple organic processes that use more
benign, or no catalysts at all.
C. Silicone Elastomer Can Benefit from
Unusual Fillers
Silicones elastomers are normally reinforced with expensive
fumed silica or really expensive MQ resins, without which
their properties are relatively poor (e.g., poor tear
resistance). We are exploring the use of other materials that
could reduce cost while improving the performance of the
silicone in use, and facilitate degradation at the end of
life. We have published on the use of alpaca wool (hence the
picture of the alpaca above, 293).
We are expanding the work to other fillers based on
saccharides (268),
and waste materials that would otherwise go to landfill.
D. Silicones Degrade Used Tire Rubber - an
Egregious, Single Use Elastomer
We reported the ability to completely dissolve used
automobile rubber under mild conditions using silicones (265).
The process is mild and efficient, but the catalyst makes the
process non-competitive on price. We are adapting the process
to use less chemistry for new applications, including
silicone elastomers reinforced with automobile rubber.

E.
New Routes to Explicit, Functional Silicones
Most silicone polymers are
complex mixtures. The Piers-Rubinsztajn reaction
(248)
allows
exquisite control over synthesis of highly
branched structures, as shown in this MDTQ
dendron (264).
These silicones will be more sustainable,
because less material will be needed for a given
application; the properties of the compounds are
explicit, rather than a composite of the
contents of the mixture. We continue to exploit
structural control over silicone to optimize
existing and target new properties.

[ return to top ]
