| Problem Set #5 | October 17, 1997 |
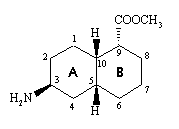
1. Construct a model of the cis-decalin compound shown below, with both the A and B rings in chairs and the two H's at 5 and 10 pointing up towards you. (The numbers included refer to the decalin numbering system.)

(a) With respect to the A-ring, say whether the following atoms or bonds are axial or
equatorial:
(i) NH2
(ii) H-5
(iii) H-10
(iv) bond 9-10
(v) bond 5-6
(br) With respect to ring B, do the same for:
(i) COOCH3
(ii) H-5
(iii) H-10
(c) Carry out a ring flip on both rings A and B (you can best do this by placing ring A in a twist-boat conformation, then B into a twist-boat, and then twisting the two rings from one twist-boat to another - you will find that the rings are now amazingly flexible - and finally going back to two chairs). Now look and see what has happened to the equatorial or axial nature of the atoms and bonds listed in (a) and (b) above. Which do you think is the more stable conformer?
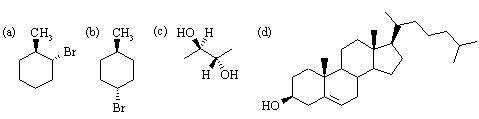
(a) (R)-2-chlorobutane
(b) (2R,3R)-3-ethyl-2-methyl-5-phenyl-1-pentanol
(c) (R)-3-fluoro-1,1-dimethylcyclohexane
(d) (E)-2-chloro-3-ethyl-3-hexene
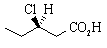 , has [
, has [
(a) What would be the observed optical rotation of a sample of 2.5 g of the compound in 100 mL of water, measured in a 40-cm tube?
(b) A sample of 3-chloropentanoic acid having [![]() ]
D25 +12.7o was recovered by resolution of a racemic mixture
of the acid. What is the enantiomeric excess of the sample recovered from the racemic mixture?
]
D25 +12.7o was recovered by resolution of a racemic mixture
of the acid. What is the enantiomeric excess of the sample recovered from the racemic mixture?
| Go to: |
Instructions for Printing this Document Answers Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
17oct97; wjl