An Introduction to the Electronic Structure of Atoms
and Molecules
Dr. Richard F.W. Bader
Professor of Chemistry / McMaster University / Hamilton,
Ontario
|
Some Chemical Implications
A detailed study of the chemical implications of
the orbital theory of electronic structure must await our discussion of
the chemical bond. However, we can at this point correlate the gross chemical
behaviour of the elements with the general results of the orbital theory.
The effective nuclear charge is a minimum for the group
I elements in any given row of the periodic table. Therefore, it requires
less energy to remove an outer electron from one of these elements than
from any other element in the periodic table. The strong reducing ability
of these elements is readily accounted for. The variation in the relative
reducing power of the elements across a given period or within a given
group will be determined by the variation in the effective nuclear charge.
The ability of the elements in a given row of the periodic table to act
as reducing agents should undergo a continuous decrease from group I to
group VII, since the effective nuclear charge increases across a given
row. Similarly, the reducing ability should increase down a given column
(group) in the table since the effective nuclear charge decreases as the
principal quantum number is increased. Anticipating the fact that electrons
can be transferred from one atom (the reducing agent) to another (the oxidizing
agent) during a chemical reaction, we expect the elements to the left of
the periodic table to exhibit a strong tendency to form positively charged
ions.
The ability of the elements to act as oxidizing agents
should parallel directly the variations in the effective nuclear charge.
Thus the oxidizing ability should increase across a given row (from group
I to group VII) and decrease down a given family. These trends are, of
course, just the opposite of those noted for the reducing ability. We can
also relate the chemical terms "reducing ability" and "oxidizing ability"
to the experimentally determined energy quantities, "ionization potential"
and "electron affinity." The reducing ability should vary inversely with
the ionization potential, and the oxidizing ability should vary directly
with the electron affinity. The elements in groups VI and VII should exhibit
a strong tendency for accepting electrons in chemical reactions to form
negatively charged ions. Francium, which possesses a single outer electron
in the 7s orbital, should be the strongest chemical reducing agent
and fluorine, with an orbital vacancy in the 2p subshell, should
be the strongest oxidizing agent. (Click
here for note.)
A great deal of chemistry can now be directly related
to the electronic structure of the elements. For example, the reaction
is explained chemically by stating that Cl2
is a stronger oxidizing agent than Br2.
The electronic interpretation is that the orbital vacancy in Cl is in a
3p orbital and closer to the nucleus than the 4p orbital
vacancy in Br. Thus the effective nuclear charge which attracts the extra
electron is larger for the Cl atom than for the Br atom. We could of course
interpret this same reaction by stating that the Br- ion is
a stronger reducing agent than is the Cl- ion. In other words
the extra electron in the Br- ion is less tightly held than
is the extra electron in the Cl- ion. The explanation in terms
of the relative effective nuclear charges is the same as that given above.
The decrease in the effective nuclear charge down the halogen
family of elements leads to some interesting differences in their chemistry.
For example, hydrogen chloride may be prepared from sodium chloride and
sulphuric acid:
| (1) |

|
However, the same method cannot be employed in the preparation of hydrogen
bromide or hydrogen iodide. In the preparation of hydrogen bromide from
sodium bromide,
| (2) |

|
some of the HBr reacts further,
| (3) |

|
and the HBr is thus contaminated. In preparation of hydrogen iodide
a further reaction again occurs:
| (4) |

|
Reactions (3) and (4)
are clearly redox reactions in which the halide ions reduce the sulphur
in the SO4-2
anion to a lower oxidation state. Since Cl has the highest effective nuclear
charge, the Cl- ion should be the weakest reducing agent of
the three halide ions. Indeed, the Cl- ion is not a strong enough
reducing agent to change the oxidation state of S in SO4-2.
The Br- ion possesses an intermediate value for the effective
nuclear charge and thus it is a stronger reducing agent than the Cl-
ion. The Br- ion reduces the oxidation number of sulphur from
(+6) to (+4). Since the I- ion binds the extra electron least
of all (the electron is in an n = 5 orbital and the effective nuclear
charge of iodine is the smallest of the three), it should be the strongest
reducing agent of the three halide ions. The I- ion in fact
reduces the sulphur from (+6) to (-2).
A word about oxidation numbers and electron density distributions
is appropriate at this point. An oxidation number does not, in general,
represent the formal charge present on a species. Thus S is not S+6
in the SO-2 ion, nor is it S-2
in the H2S molecule. However, the average
electron density in the direct vicinity of the sulphur atom does increase
on passing from SO4-2
to H2S. From their relative positions in
the periodic table it is clear that oxygen will have a greater affinity
for electrons than sulphur. Thus when sulphur is chemically bonded to oxygen
the electron density in the vicinity of the sulphur atom is decreased over
what it was in the free atom and increased in the region of each oxygen
atom. Again it is clear from the relative positions of H and S in the periodic
table that sulphur has a greater affinity for electrons than does hydrogen.
Thus in the molecule H2S, the electron
density in the vicinity of the sulphur atom is increased over that found
in the free atom. In changing the immediate chemical environment of the
sulphur atom from that of four oxygen atoms to two hydrogen atoms, the
electron density (i.e., the average number of electrons) in the vicinity
of the sulphur atom has increased. The assignment of actual oxidation numbers
is simply a bookkeeping device to keep track of the number of electrons,
but the sign of the oxidation number does indicate the direction of the
flow of electron density. Thus sulphur has a positive oxidation number
when combined with oxygen (the sulphur atom has lost electron density)
and a negative one when combined with hydrogen (the electron density around
sulphur is now greater than in the sulphur atom).
The above are only a few examples of how a knowledge
of the electronic structure of atoms may be used to understand and correlate
a large amount of chemical information. It should be remembered, however,
that chemistry is a study of very complex interactions and the few simple
concepts advanced here cannot begin to account for the incredible variety
of phenomena actually observed. Our discussion has been based solely on
energy, and energy alone never determines completely the course of a reaction
on a macroscopic level, i.e., when many molecules undergo the reaction.
There are statistical factors, determined by the changes in the number
of molecules and in the molecular dimensions, which must also be considered.
Even so, the energy effect can often be overriding.
In the long form of the periodic table, families are labelled
by both a number and by the letter A or B. Thus there is a IA family and
a IB family. It will be noted that the elements in a B family all occur
in the series of transition elements in which the d orbitals are
being filled. In the A families, however, the d orbitals are either
absent or are present as closed inner shells. For example, consider the
electronic configurations of K (IA) andCu (IB):
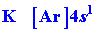
|

|
Note that the most stable configuration for Cu is not [Ar] 3d94s2
as expected. By transferring one of the 4s electrons to the 3d
vacancy, the d subshell is filled and the electronic energy is lowered.
The electron density distribution of the Cu atom is therefore a spherical
one. Both K and Cu have one outer electron with a spherical charge distribution.
They should have some properties in common, such as a tendency to lose
one electron and form a positive ion. For this reason both families are
labelled I. However, the shell underlying the outer electron in the K atom
possesses a rare gas configuration, while in the Cu atom it is a set of
filled d orbitals. This difference in electronic structure is sufficient
to cause considerable differences in their chemistry, hence the further
labels A and B.
A rare gas configuration is always one of great stability,
particularly when it occurs in a positive ion. (Recall that I2
= 47.3 ev for sodium.) The species K+2
is never observed in solution chemistry, and could be produced in the gas
phase only by an expenditure of energy far in excess of that observed in
ordinary chemical reactions. The Cu+ ion,
on the other hand, very readily loses a second electron to form the Cu+2
ion. Indeed, Cu+2 is the more common ionic
form of copper. Thus the d10 closed
shell structure is more easily broken than a rare gas configuration, giving
to Cu a variable valency of one or two.




