An Introduction to the Electronic Structure of Atoms and Molecules
Professor of Chemistry / McMaster University / Hamilton, Ontario
An Introduction to the Electronic Structure of Atoms and MoleculesProfessor of Chemistry / McMaster University / Hamilton, Ontario
|
The distribution of negative charge in a molecule will exhibit varying degrees of asymmetry depending on the relative abilities of the nuclei in the molecule to attract and bind the electronic charge density. The symmetry or asymmetry of the charge distribution plays a fundamental role in determining the chemical properties of the molecule and consequently this property of the charge distribution is used as a basis for the classification of chemical bonds.
We can envisage two extremes for the distribution of the valence charge density. An example of one of the extremes is obtained when a bond is formed between two identical atoms. The charge density of the valence electrons will in this case necessarily be delocalized equally over corresponding regions of each nucleus since both nuclei will attract the electron density with equal force. Such an equal sharing of the charge density is an example of covalent binding and is exemplified by the molecular charge distribution of N2 (Fig. 7-1).
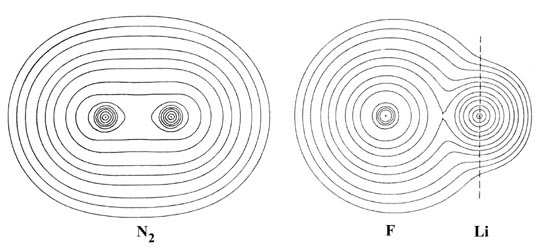
The charge distribution of LiF (Fig. 7-1) provides an example of the other extreme, termed ionic binding, obtained when a bond is formed between two atoms with very different affinities for the electronic charge density. The very unsymmetrical distribution of charge in LiF is not simply a reflection of the fluorine atom possessing seven valence electrons to lithium's one. Instead the formation of the bond in LiF corresponds to the nearly complete transfer of the valence charge density of lithium to fluorine resulting in a molecule best described as Li+F-. We need only recall that initially a lithium atom is considerably larger than a fluorine atom to realize that a considerable transfer of charge has occurred in the formation of the LiF molecule.
In N2 the valence charge density is delocalized over the whole molecule. The electronic charge is heavily concentrated in the internuclear region where it forms a bridge of high density between the two nuclei. Only the density of the 1s inner shell or "core" orbitals is strongly localized in the regions of the nuclei. In contrast to this, practically all of the charge density in the lithium fluoride molecule is localized in nearly spherical contours on the two nuclei in the manner characteristic of two separate closed-shell distributions. Only contours of very small value encompass both nuclei and the bridge of charge density joining the two spherical distributions is very low in value, being approximately one tenth of the value observed for N2.
We may determine the total amount of electronic charge
in an arbitrary region of space by summing the density in each small volume
element within the region of interest (i.e., integrating the charge distribution
over some particular volume of space):
|
|
The nonbonded charge density of the lithium nucleus in LiF is 1.07 e- compared to 1.5 e- in the Li atom (i.e., one half of the total number of electronic charges in a Li atom). The nonbonded charge density of the F nucleus, on the other hand, is increased above its atomic value, being 5.0 e- as compared to 4.5 e- in the fluorine atom. Since the distributions centred on the nuclei in LiF are nearly spherical, the total charge contained in each distribution will be approximately twice the value of the corresponding nonbonded charge. The total distribution of charge in LiF is, therefore, consistent with the ionic model Li+F- corresponding to the transfer of the single 2s valence electron of lithium to the fluorine. The radii of the nonbonded charge distributions (the distance measured along the bond axis from a nucleus to the outermost contour of its nonbonded density) are also consistent with the ionic model. The radius of the nonbonded charge density on lithium is 1.7 ao, a value almost identical to the radius of a Li+ ion (1.8 ao) but much less than the radius of a Li atom (3.3 ao). The value of 3.0 ao for the radius of the nonbonded charge density on fluorine is consistent with that of a fluoride ion distribution as it represents a slight increase over the atomic value for fluorine of 2.8 ao.
By way of comparison, the nonbonded charge on the nitrogen nuclei in N2 is increased above the atomic value of 3.5 e- to 3.68 e-. This transfer of charge density to the nonbonded regions on bond formation is somewhat surprising when it is recalled that charge density must be accumulated in the binding region, the region between the nuclei, to achieve a chemical bond. We require a more detailed picture of the charge reorganization accompanying the formation of a bond to understand fully the distribution of the charge density in a molecule. In addition, many chemical bonds possess charge distributions which lie between the extreme of the perfect sharing of the valence charge density found in N2 and its complete localization on one nucleus in LiF.
We consider next a method of classification of bonding in molecules, a classification which provides at the same time an understanding of the mechanism of the two binding situations in terms of the forces exerted on the nuclei.
 |
 |
 |
 |